
The problem with this definition is that vague terms, such as comparison and examination of association, might be interpreted as an analytic comparison of at least two exposures (i.e., interventions, risk factors or prognostic factors).
#Retrospective cohort study vs case control series
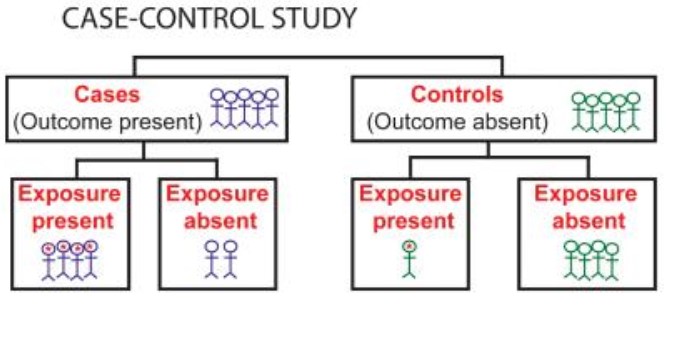
The only difference between cohort studies and case series in many definitions is that cohort studies compare different groups (i.e., examine the association between exposure and outcome), while case series are uncontrolled. The lack of a comparison group is of particular importance for distinguishing cohort studies from case series because in many definitions, they share a main design feature of having a follow-up period examining the exposed individuals over time. One key design feature to classify observational study designs is to distinguish comparative from non-comparative studies. However, the distinction between different non-randomized study designs is difficult.

Systematic reviews that include non-randomized studies often consider different observational study designs. On the other hand, the workload would increase (e.g., additional data extraction and risk of bias assessment, as well as reanalyses). Therefore, on the one hand, the outlined approach can increase the confidence in effect estimates and the strengths of conclusions. Consequently, more studies could be included in a systematic review. There are possibly large numbers of studies without a comparison for the exposure of interest but that do provide the necessary data to calculate effect measures for a comparison. Instead, all studies for which sufficient data are available for reanalysis to compare different exposures (e.g., sufficient data in the publication) are classified as cohort studies. The term “enables/can” means that a predefined analytic comparison is not a prerequisite (i.e., the absolute risks per group and/or a risk ratio are provided). We discuss the potential impact of the proposed conceptualization on the body of evidence and workload.Īll studies with exposure-based sampling gather multiple exposures (with at least two different exposures or levels of exposure) and enable calculation of relative risks that should be considered cohort studies in systematic reviews, including non-randomized studies. The main aim of this conceptualization is to clarify the distinction between cohort studies and case series. We propose a conceptualization of cohort studies in systematic reviews of comparative studies. Distinguishing cohort studies from case series is difficult.

